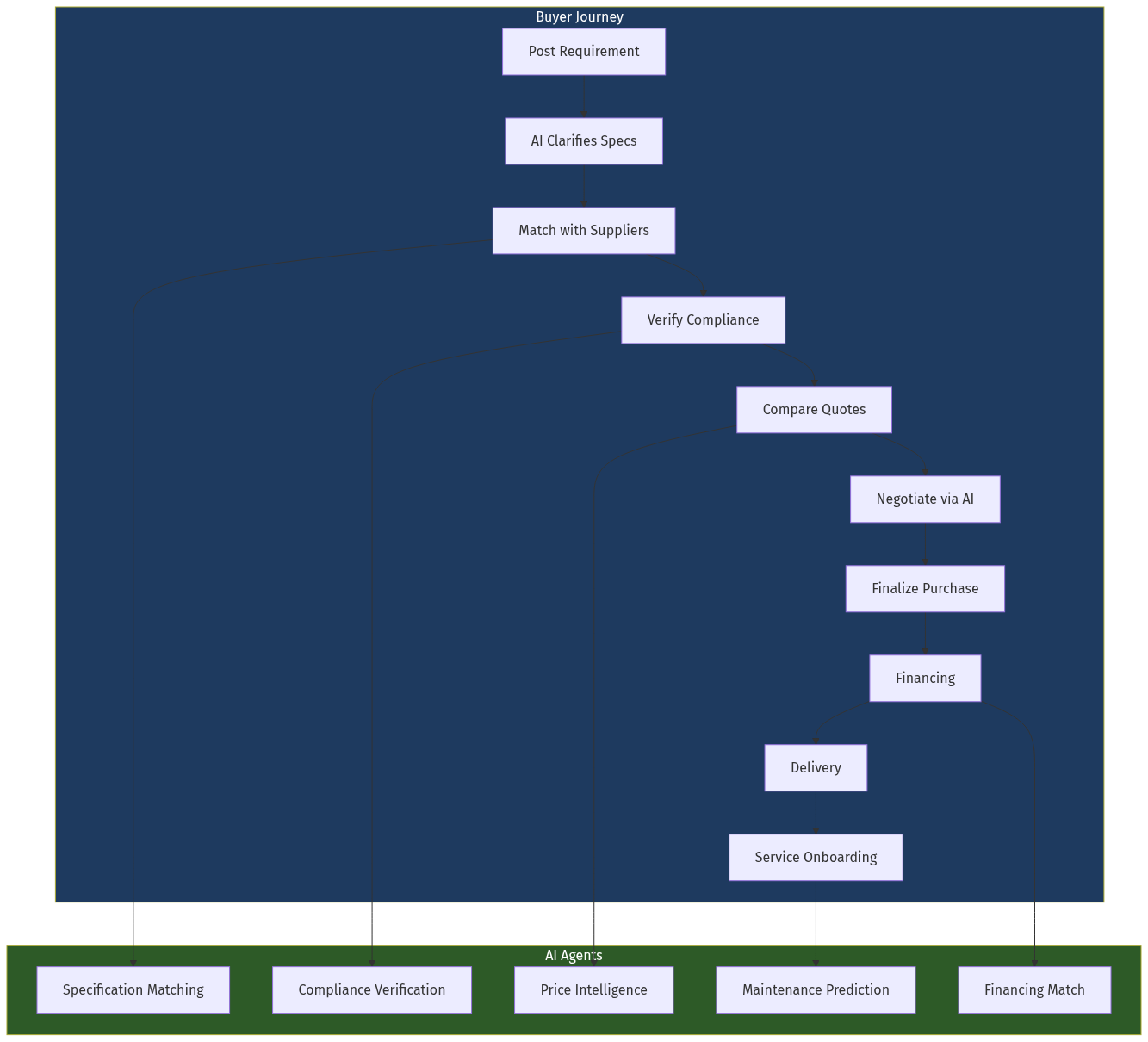
Gap 1: No Specialized Marketplace
General B2B platforms (IndiaMART, TradeIndia) list cold chain equipment but without specialization—no temperature compliance filters, no pharmaceutical-grade verification, no real-time availability.
Gap 2: Specification Chaos
Equipment specifications are inconsistent across manufacturers. No standard format for:
- Temperature range capabilities
- Compressor types and efficiency ratings
- Energy consumption metrics
- Compliance certifications (FDA, GMP, BIS)
Gap 3: Price Opacity
Same equipment can have 20-30% price variance between dealers. No transparent pricing mechanism exists.
Gap 4: Compliance Verification
Pharma companies need FDA, WHO-GDP, and BIS certified equipment. Currently, verification requires manual certificate checking—prone to fraud.
Gap 5: Service Fragmentation
After-sales service is dealer-dependent. No unified service marketplace with:
- Technician availability
- Spare parts inventory
- Maintenance contracts
- Emergency repair guarantees
Gap 6: Financing Gap
Equipment finance for cold chain is limited. No integrated financing marketplace with:
- Leasing options
- EMI calculators
- Pre-approved financing
- Tax benefit calculators