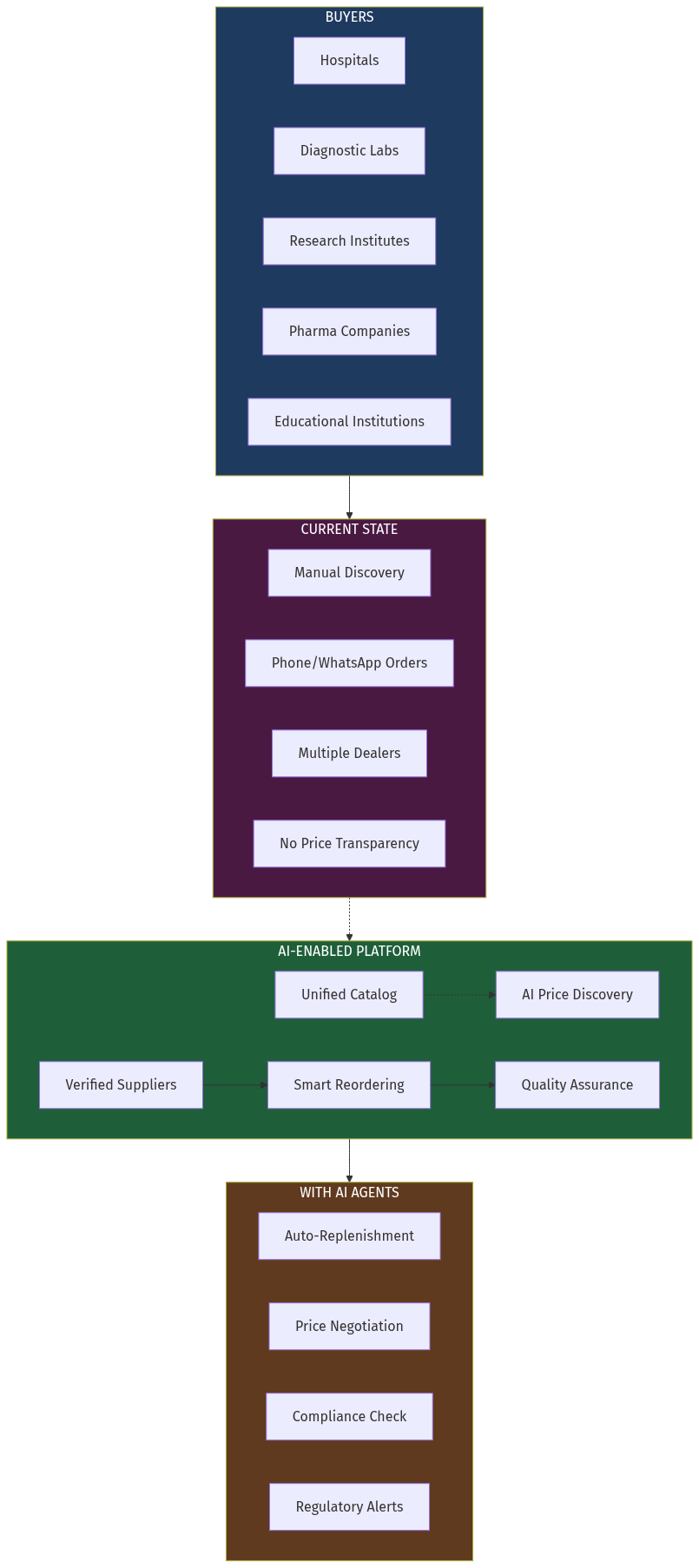
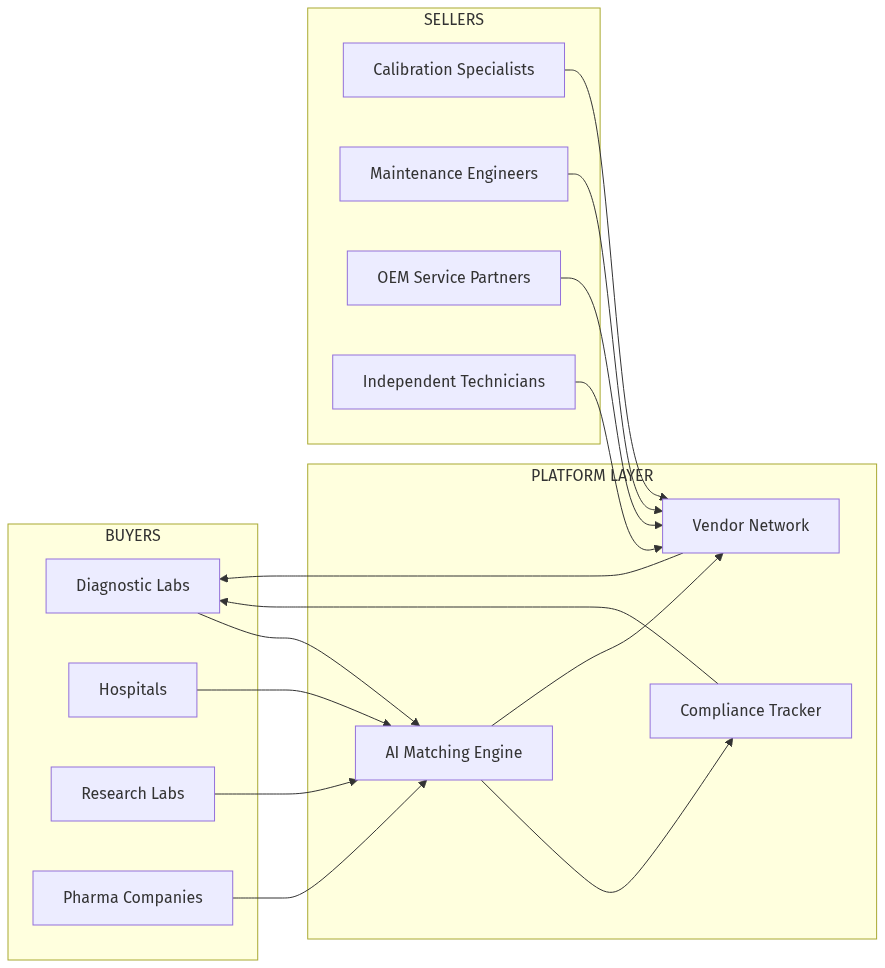
Laboratory equipment calibration is a $12 billion global market characterized by extreme fragmentation. When a hospital's blood analyzer needs calibration, the biomedical engineering team must identify certified vendors, compare quotes, schedule service, and manually track compliance documentation — a process that takes 2-4 weeks for routine calibrations and much longer for specialized equipment.
The core insight: Equipment-calibration matching is fundamentally a structured data problem. AI can parse equipment specifications, match them to vendor certifications, verify regulatory compliance, and schedule services — work that takes procurement teams days or weeks. Why now:- Vision-language models can read equipment nameplates and serial numbers from photos
- Embedding models understand equipment-certification relationships semantically
- Agent architectures can coordinate multi-vendor scheduling
- Regulatory pressure (NABH, NABL, FDA) is increasing compliance requirements
- India's diagnostic market is growing 20%+ annually