Every manufacturing facility lives in constant fear of three words: "Failed Audit Results." A single FDA, OSHA, or ISO non-compliance finding can trigger shutdowns costing $50,000–$500,000 per day. Yet facilities juggle 50+ simultaneous regulatory frameworks, generate thousands of pages of documentation per audit, and rely on overloaded compliance officers working 60-hour weeks just to stay afloat.
The current state: Fragmented spreadsheets, disconnected document systems, reactive firefighting, and a perpetual state of audit anxiety.
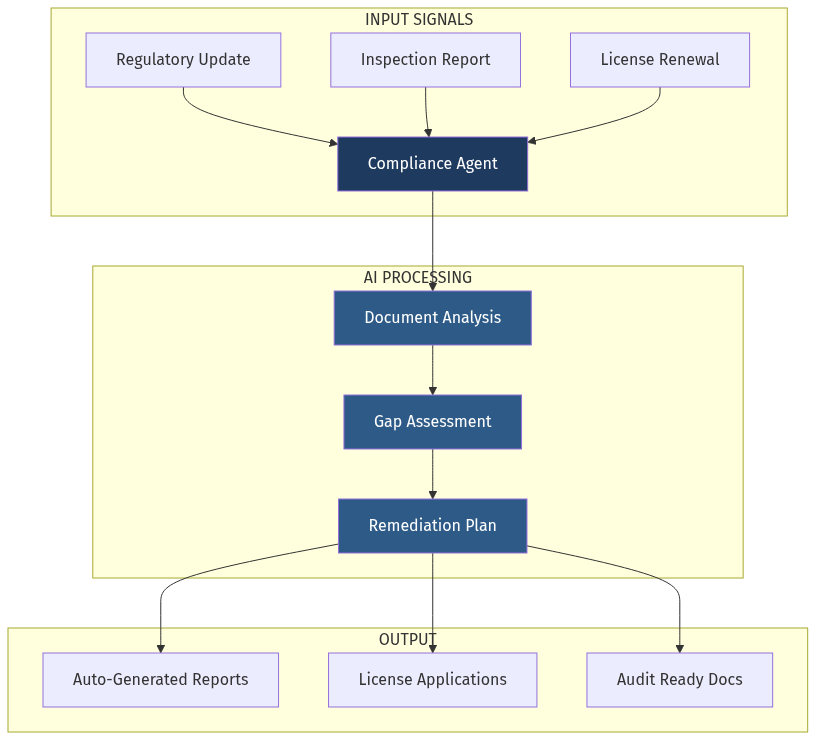
The opportunity: An AI-powered compliance automation platform that:
- Continuously monitors regulatory changes across all jurisdictions
- Auto-generates required documentation from existing operational data
- Conducts real-time gap assessments against upcoming inspections
- Prepares audit-ready packages in minutes, not weeks
- Learns from every inspection to predict and prevent the next finding